
Trastuzumab deruxtecan (Enhertu®) is an innovative targeted therapy used to treat certain secondary (metastatic, advanced) breast cancer patients. In this blog we will delve into the science of trastuzumab deruxtecan while looking at a patient’s experience on this drug. This blog was written in collaboration with Make2ndsCount.
What is trastuzumab deruxtecan (Enhertu)?
Trastuzumab deruxtecan is a type of targeted therapy called an antibody-drug conjugate (ADC). ADCs are a novel type of therapy that combines highly specific monoclonal antibodies with powerful chemotherapy drugs 1. In this case, trastuzumab deruxtecan is made up of the monoclonal antibody trastuzumab (Herceptin®) linked to the chemotherapy drug deruxtecan (figure 1).
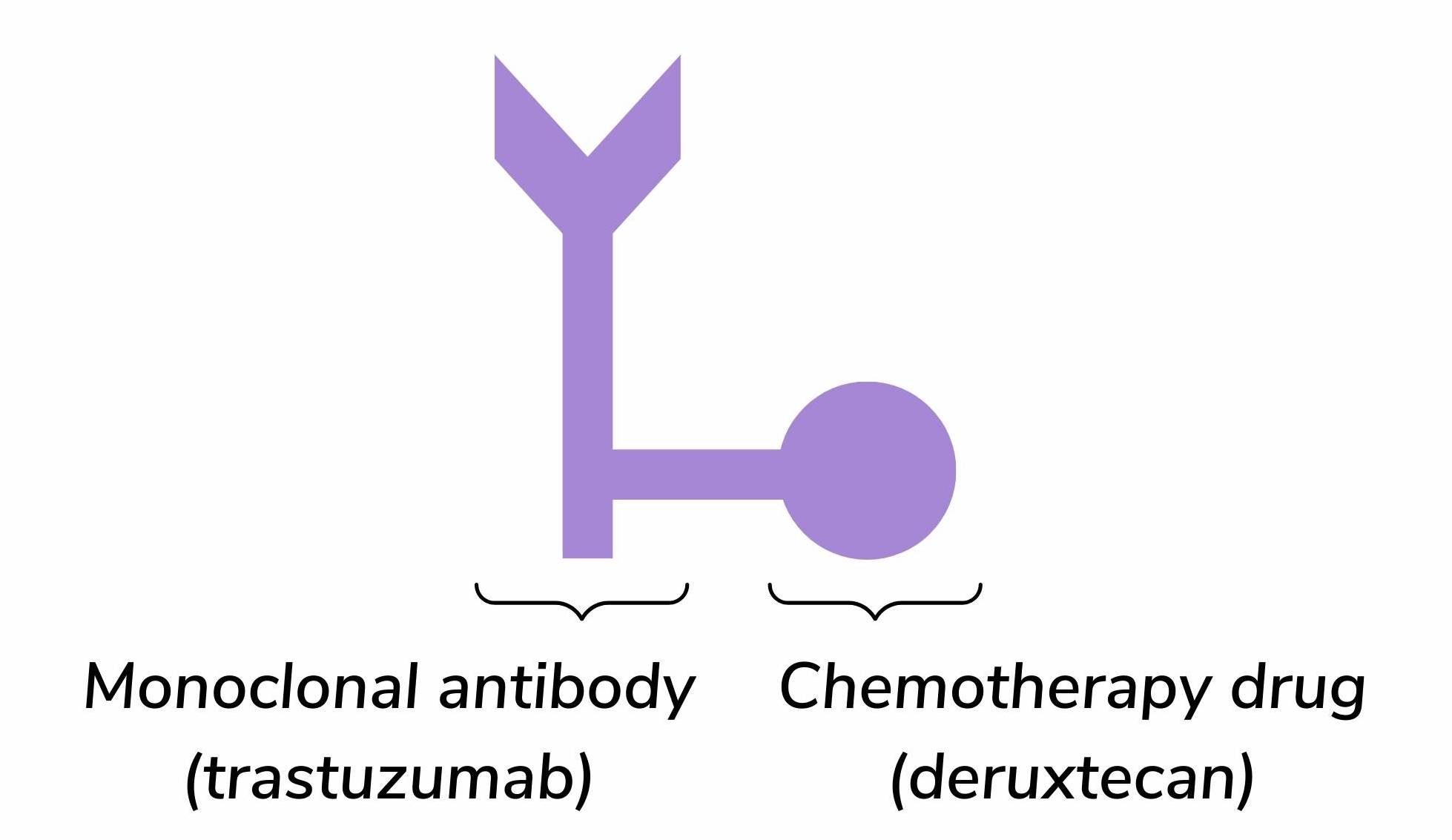
Figure 1. The structure of trastuzumab deruxtecan (Enhertu).
How does trastuzumab deruxtecan (Enhertu) work?
The trastuzumab monoclonal antibody component specifically targets and binds to human epidermal growth factor receptor 2 (HER2) proteins that is present in high amounts of the surface of cancer cells. Upon binding of trastuzumab to HER2 the chemical linker is split by enzymes at the site of the cancer cells, releasing the deruxtecan chemotherapy2. Deruxtecan is then taken in by cancer cells and prevents them from growing and dividing, resulting in cell death. This highly targeted mechanism is designed to minimise damage to healthy cells, improve the impact of the drug and reduce side effects3.
Who is trastuzumab deruxtecan (Enhertu) for?
As trastuzumab deruxtecan targets cells with high levels of HER2 on the surface, it is a treatment option for some patients with cancer that expresses the HER2 protein.
For secondary HER2-positive breast cancer
Trastuzumab deruxtecan is approved to treat advanced HER2-positive (HER2+) breast cancer in the UK, US and EU4,5,6. It is currently a second line treatment option for people who have received previous treatment for secondary HER2+ breast cancer that was unsuccessful.
HER2+ breast cancer is defined by an immunohistochemistry (IHC) test score of 3+. Breast cancer is also considered HER2+ with a IHC score of 2+ combined with a positive fluorescence in situ hybridisation (FISH) test.
For secondary HER2-low breast cancer
More recently, trastuzumab deruxtecan was approved to treat HER2-low breast cancer patients in the US and EU6,7. HER2-low is a new way to classify breast cancer, it includes some hormone receptor-positive (HR+) and triple negative breast cancers (TNBC) that were previously considered HER2-negative (HER2-). Patients must have received prior chemotherapy to treat metastatic breast cancer or developed a recurrence during or within six months of completing chemotherapy after surgery (adjuvant treatment) to be eligible for treatment.
HER2-low breast cancer is a relatively new concept that is defined by an IHC score of 1+ or IHC+ with a negative FISH test. Research suggests that up to 55% of breast cancers are now considered HER2-low, making trastuzumab deruxtecan a potential treatment option for many secondary breast cancers8.
What clinical evidence is there for trastuzumab deruxtecan (Enhertu)?
Clinical evidence in metastatic HER2-positive breast cancer: DESTINY-Breast03
The DESTINY-Breast03 trial looked at trastuzumab deruxtecan in the HER2+ population and compared trastuzumab deruxtecan with trastuzumab emtansine (Kadcyla®) in 524 patients previously treated with trastuzumab (Herceptin) and chemotherapy. Recently published follow-up results demonstrated that for most patients on trastuzumab deruxtecan the cancer did not grow or spread for 28.8 months, compared to 6.8 months on trastuzumab emtansine9. Survival results were also published, which found that 77.4% of patients on trastuzumab deruxtecan were alive at 24 months, compared to 69.9% on trastuzumab emtansine9.
Clinical evidence in metastatic HER2-low breast cancer: DESTINY-Breast04
The DESTINY-Breast04 trial looked at trastuzumab deruxtecan in the HER2-low population and compared trastuzumab deruxtecan with chemotherapy. This trial consisted of 540 patients, 480 of these patients had hormone receptor-positive (HR+) breast cancer and 60 had triple negative breast cancer (TNBC). The results showed that for most patients the cancer did not grow or spread for 9.9 months while on trastuzumab deruxtecan, compared to 5.1 months while on chemotherapy10.
These results varied depending on the subtype of breast cancer. For HR+ patients, most went on without the cancer growing or spreading for 10.1 months on trastuzumab deruxtecan, compared to 5.4 months on chemotherapy10. For TNBC patients these figures were 8.5 months on trastuzumab deruxtecan, compared to 2.9 months on chemotherapy10.
It was also found that most patients on trastuzumab deruxtecan lived 23.4 months longer on averaged, compared to 16.8 months on chemotherapy10. Those with HR+ breast cancer on trastuzumab deruxtecan lived 23.9 months longer on average, compared to 17.5 months on chemotherapy10. For TNBC patients, those on trastuzumab deruxtecan lived 18.2 months longer on average, compared to 8.3 months on chemotherapy10.
What are the side effects of trastuzumab deruxtecan (Enhertu)?
While trastuzumab deruxtecan offers remarkable benefits, like all medications, it may come with potential side effects. Common side effects include11:
- Fatigue
- Nausea
- Hair loss
- Diarrhoea
It may also cause more serious side effects and it is important to be aware of the signs and symptoms11:
- Interstitial lung disease (ILD) – Cough, trouble breathing, shortness of breath, fever, new or worsening breathing symptoms.
- Heart problems – New or worsening shortness of breath, cough, feeling tired, swelling in legs or ankles, irregular heartbeat, sudden weight gain, feeling dizzy, lightheaded or loss of consciousness.
- Reduced white blood cell count (neutropenia) – Signs of infection, fever, chills.
Have you tried tracking your side effects on OWise? With our easy-to-use sliders, you can track over 30 different side effects and visualise how your trends change over time. This makes monitoring symptoms easy and you can even share them with your care team.
How are serious side effects of trastuzumab deruxtecan (Enhertu) managed?
ILD can be managed with medications such as corticosteroids, medication that reduce stomach acid and medications that slow the progression of lung scarring12. Heart conditions can be monitored by tests such as echocardiography, CT or PET scans, and can be managed by pausing trastuzumab deruxtecan treatment11,13. Neutropenia can be managed by regular monitoring of white blood cell counts11. If necessary, there are treatment options available that can increase white blood cell count14. In the case of infection, antibiotics, white blood cell transfusions and stem cell transplants are possible courses of action.
Jenny Read’s Story: Diagnosis to trastuzumab deruxtecan (Enhertu)
I am tolerating the treatment reasonably well – tiredness is the biggest issue, some days I need to stay in bed all day.
Read more about Jenny’s story below
What is your secondary breast cancer diagnosis background?

I was first diagnosed with HER2+ breast cancer in February 2017 when I was 38, then it was discovered that it had already spread to my liver and spine the following week. This all happened 8 weeks after my daughter was born. I have two children: a son now aged 10, and a daughter now aged 6.
I was reassured it was most likely a blocked milk duct when I queried a lump I found at my 38 week antenatal appointment. I queried this again at my 6-week post-natal check, by which time my nipple had become inverted, and was urgently referred to the breast clinic by my GP.
What treatments have you been on up until trastuzumab deruxtecan (Enhertu)?
Initial Treatments (2017-2020)
After a single high dose of radiotherapy on my back the first treatment I had was docetaxel (Taxotere®) along with Herceptin. My hair of course fell out, but showers were a lot quicker…every cloud and all that. The multiple lesions on my liver reduced, although it remained inoperable.
I had 8 rounds of chemo but was not offered a mastectomy due to the secondary diagnosis. However, after the appearance of the tumour changed on a scan, I did end up getting a left mastectomy in August 2018. Although doing nothing to affect my prognosis, it was a mental relief to have the tumour removed from my body.
I remained on 3-weekly Herceptin, tamoxifen and goserelin (Zoladex®) until I decided to have my ovaries removed in May 2019. This was more to reduce the number of medical appointments I had to attend, as I felt that it was becoming too much to bear. I was then moved to letrozole (Femara®), and of course the Zoladex stopped. This treatment regime continued until July 2020, until the headaches started.
Brain Surgery and Radiotherapy (2020-2021)
I’ve never been one to contact the breast care nurses much, so when I did, they moved quickly and got me in for a CT scan the following day. I was immediately called back and told to report to The Victoria Hospital in Kirkcaldy ASAP – I had a large tumour on the front right of my brain. I was put on to a course of high dose steroids (dexamethasone) to try and reduce the swelling, and told not to drive. I was transferred to the Royal Infirmary of Edinburgh, where the Neurological team had just moved from The Western General, for brain surgery.
This happened on Friday 13th August 2020. Yes, you read that right…I had open brain surgery on Friday 13th! I figured that as my son was born on the 13th July, it must be a lucky number for me! Recovery was reasonably quick, and I was back home the following Tuesday.
The first scan results showed that vast majority of the tumour had been removed, although I’d still need whole brain radiotherapy to sweep up the tentacle like remnants. This resulted in me losing my hair again and severe tiredness. I then reverted to my previous 3-weekly Herceptin treatment.
HER2CLIMB-02 Clinical Trial (2021-2023)
Once recovered, my oncologist informed me of a trial at The Western called HER2CLIMB-02 and asked if I would like to participate in it. This trial compared trastuzumab emtansine (Kadcyla) with tucatinib vs Kadcyla with placebo in HER2+ secondary breast cancer patients15.
I started this in September 2021, with treatment every 3 weeks. My time on the trial ended after 18 months after very small progression unfortunately exceeded the maximum % growth permitted for trial parameters. The code was unlocked and I was found to be on placebo and not the trial drug. I wasn’t too disappointed as I had still been receiving chemo in the form of Kadcyla, so I didn’t think of it as a waste of my time.
What has been your experience on trastuzumab deruxtecan (Enhertu)?
From April 2023 it was on to Enhertu. Up to the present day (25th August 2023) I have had 7 treatments with a brain MRI scan showing reduction, with some areas disappearing completely.
Have you had side effects while on trastuzumab deruxtecan (Enhertu)?
Once again, I am tolerating the treatment reasonably well – tiredness is the biggest issue, some days I need to stay in bed all day. I have two very demanding young children and have (just!) survived the school holidays, so I’ll hopefully begin to feel better with some more peace and quiet to rest. My hair is definitely thinning, although it was so thick beforehand that it’s barely noticeable but some patches are appearing now. I’m given granisetron anti-sickness tablets to take for 4 days after infusions. Although they have kept the sickness at bay, I do suffer from constipation but Laxido helps me cope with that.
Do you think the OWise app can benefit secondary breast cancer patients?
Unfortunately, I have not used the OWise app throughout my treatment.
After downloading the OWise app recently, I can see the benefits of logging treatment information in the one location.
Something I struggled with was fitting in everyday tasks around my numerous medical appointments and setting aside some “me” time. That was behind my decision to have my ovaries removed, after counting up over 80 medical appointments on the calendar.
And that’s trastuzumab deruxtecan (Enhertu) summed up
Trastuzumab deruxtecan is a remarkable advancement in targeted therapy for secondary breast cancer patients. Its impressive clinical data and approval for use in advanced HER2+ and HER2-low (US only) breast cancer offers hope for patients who have exhausted other treatment options. Side effects may occur, but open communication with the care team can help patients to manage these challenges and improve outcomes.
At OWise, we want to make sure you are kept informed so make sure to follow our Instagram and Twitter for any updates. Any questions? Get in touch!
References
- Pettinato MC. Introduction to Antibody-Drug Conjugates. Antibodies. 2021;10(4):42.
- Enhertu : EPAR – Product Information. 2021. Available at: https://www.ema.europa.eu/en/documents/product-information/enhertu-epar-product-information_en.pdf.
- Hofland P, Portillo S. What are Antibody-drug Conjugates? ADC Review / Journal of Antibody-drug Conjugates. 2019.
- Trastuzumab deruxtecan for treating HER2-positive unresectable or metastatic breast cancer after 1 or more anti-HER2 treatments. 2023. Available at: https://www.nice.org.uk/guidance/ta862.
- FDA grants regular approval to fam-trastuzumab deruxtecan-nxki for breast cancer. 2022. Available at: https://www.fda.gov/drugs/resources-information-approved-drugs/fda-grants-regular-approval-fam-trastuzumab-deruxtecan-nxki-breast-cancer.
- Enhertu. 2021. Available at: https://www.ema.europa.eu/en/medicines/human/EPAR/enhertu
- FDA approves first targeted therapy for HER2-low breast cancer. 2022. Available at: https://www.fda.gov/news-events/press-announcements/fda-approves-first-targeted-therapy-her2-low-breast-cancer
- Li Y, Julia, Tam FY, Thomson Loong, Tse GM. Comprehensive characterization of HER2-low breast cancers: implications in prognosis and treatment. EBioMedicine. 2023;91:10457
- Stenger M. Updated Results of DESTINY-Breast03 T-DXd vs T-DM1 for Previously Treated Patients With HER2-Positive Metastatic Breast Cancer – The ASCO Post. 2023. Available at: https://ascopost.com/news/january-2023/updated-results-of-destiny-breast03-t-dxd-vs-t-dm1-for-previously-treated-patients-with-her2-positive-metastatic-breast-cancer/.
- Modi S, Jacot W, Yamashita T, Sohn J, Vidal M, Tokunaga E, Tsurutani J, Ueno NT, Prat A, Chae YS, et al. Trastuzumab Deruxtecan in Previously Treated HER2-Low Advanced Breast Cancer. New England Journal of Medicine. 2022;387(1).
- AstraZeneca, Daiichi-Sankyo. Potential Risks of Treatment | ENHERTU® (fam-trastuzumab deruxtecan-nxki). Available at: https://www.enhertu.com/en/breast/risks-and-side-effects
- Mayo Clinic. Interstitial lung disease – Diagnosis and treatment – Mayo Clinic. www.mayoclinic.org. 2023. Available at: https://www.mayoclinic.org/diseases-conditions/interstitial-lung-disease/diagnosis-treatment/drc-20353113#:~:text=Many%20people%20diagnosed%20with%20interstitial
- Breast Cancer. Enhertu. www.breastcancer.org. 2022. Available at: https://www.breastcancer.org/treatment/targeted-therapy/enhertu
- American Cancer Society. Low White Blood Cell Counts | Neutropenia. www.cancer.org. 2022. Available at: https://www.cancer.org/cancer/managing-cancer/side-effects/low-blood-counts/neutropenia.html
- Curigliano G. HER2CLIMB-02 Trial to Assess Tucatinib and T-DM1 in HER2+ Breast Cancer. Targeted Oncology. 2023. Available at: https://www.targetedonc.com/view/her2climb-02-trial-to-assess-tucatinib-and-t-dm1-in-her2-breast-cancer
